Bioprocess Validation Market Size to Surge USD 1,179.55 Million by 2034 with a CAGR of 9.13%
The global bioprocess validation market is projected to reach an impressive USD 1,179.55 million by 2034, expanding at a compound annual growth rate (CAGR) of 9.13% from 2025. Driven by the rapid adoption of advanced biomanufacturing technologies, including cell and gene therapies, and the increasing emphasis on automation, real-time analytics, and integrated validation solutions, this market underscores the crucial role of validation in ensuring product safety, efficacy, and regulatory compliance in the biotech and pharmaceutical sectors.
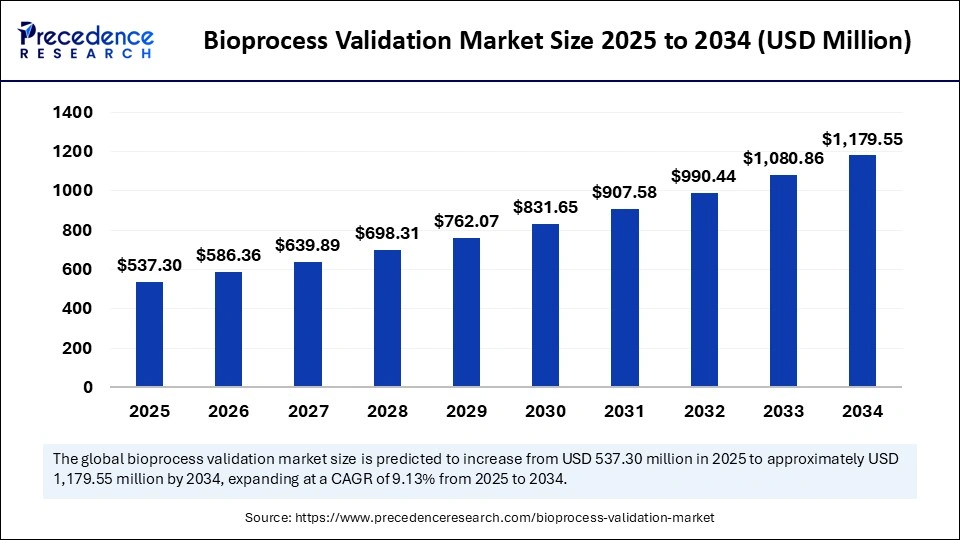
Bioprocess Validation Market Key Insights
-
The market was valued at USD 537.3 million in 2025 and is expected to nearly double by 2034.
-
North America holds the largest market share of 39% in 2024.
-
Asia Pacific is projected to experience the fastest growth, with a CAGR of over 9%.
-
Major players include Thermo Fisher Scientific, Merck KGaA, and Sartorius AG.
-
Microbiological testing dominates with a 34.4% share, reflecting the critical need for sterility in biologics.
The Role of AI in Bioprocess Validation
Artificial intelligence is transforming bioprocess validation through automated, real-time data analysis and process optimization. AI-driven systems leverage sensors, digital twins, and machine learning models to detect deviations early, ensuring continuous product quality throughout manufacturing.
AI’s integration into validation workflows accelerates decision-making, reduces human error, and enhances compliance by enabling predictive analytics and automating repetitive tasks. This digital evolution supports biopharmaceutical companies in achieving faster regulatory approvals and lowering manufacturing costs, fueling market growth.
What Drives Market Expansion?
The surge in biopharmaceutical R&D, rising demand for personalized medicine, and stringent regulatory standards are key growth drivers. Additionally, the shift towards digital bioprocessing, the adoption of single-use systems, and increasing outsourcing to specialized contract manufacturing organizations (CMOs) further bolster the market.
Bioprocess Validation Market Scope
| Report Coverage | Details |
| Market Size in 2025 | USD 537.30 Million |
| Market Size in 2026 | USD 586.36 Million |
| Market Size by 2034 | USD 1,179.55 Million |
| Market Growth Rate from 2025 to 2034 | CAGR of 9.13% |
| Dominating Region | North America |
| Fastest Growing Region | Asia Pacific |
Opportunities and Trends
What are the emerging opportunities in bioprocess validation?
The adoption of digital twin technology, combined with AI and IoT, presents opportunities for predictive validation and continuous monitoring, reducing downtime and batch failures. The move toward automation in validation processes and the rising demand for validation services in emerging economies like China and India open avenues for new entrants and established players.
Regional & Segmentation Analysis
North America remains the dominant region due to robust investment in biopharmaceuticals, stringent regulations, and advanced manufacturing infrastructure. Meanwhile, Asia Pacific is experiencing rapid growth, driven by government initiatives and expanding biologics manufacturing hubs.
Segmentation by test types highlights microbiological testing as the largest segment, emphasizing sterility’s importance. The filtration validation segment leads the process component landscape, reflecting its critical role in bioprocess safety.
Bioprocess Validation Market Challenges and Cost Pressures
Despite the promising growth, challenges include high validation costs, evolving regulatory standards, and technological complexity. Manufacturers must balance investment in cutting-edge validation solutions with cost containment.
Segment Insights
Test Type Insights
Microbiological Testing Leads the Market
In 2024, the microbiological testing segment dominated the bioprocess validation market with a 34.4% share due to the high risk of microbial contamination in biopharmaceuticals such as vaccines, gene therapies, and antibodies. Since these products are derived from living cells, contamination at any stage can cause batch failures or life-threatening infections. Meanwhile, the extractables & leachables (E&L) testing segment is projected to record the fastest growth (CAGR 9.0%) owing to the surge in single-use bioprocessing systems and stricter regulatory standards.
Process Component Insights
Filters Segment Maintains Market Dominance
The filters segment held the largest market share of 36.4% in 2024, as filtration ensures sterility and product purity—crucial for regulatory compliance. Increasing production of complex biologics and the integration of single-use technologies have reinforced the importance of filter validation. The media containers & bags segment is projected to grow at a CAGR of 8.8%, driven by the rising use of disposable systems that minimize contamination and lower infrastructure costs.
Stage of Bioprocess Insights
Upstream Bioprocessing Takes the Lead
The upstream bioprocessing segment accounted for 52.1% of the market in 2024, driven by its critical role in defining cell lines, media, and bioreactor parameters that influence downstream quality. Proper validation at this stage mitigates financial and quality risks. The fill-finish operations segment is expected to expand at a CAGR of 8.9%, fueled by regulatory stringency and advanced aseptic technologies like pre-filled syringes and automated systems.
End-User Insights
Biotechnology & Pharmaceutical Companies Dominate
Biotechnology and pharmaceutical companies held a 52.1% share in 2024, leading due to their focus on producing complex biologics, biosimilars, and advanced therapies that require stringent process validation to meet FDA and EMA standards. The CDMOs segment is anticipated to grow at the fastest CAGR of 9.6%, supported by outsourcing trends, scalability needs, and expertise in handling complex biologics.
Process Type Insights
Validation of Filtration Systems Leads the Market
The validation of filtration systems segment led with a 48.5% share in 2024, underscoring its essential role in ensuring the sterility and safety of biopharmaceuticals. Rising demand for biologics and evolving regulations have made robust filter validation indispensable. The cleaning validation segment is forecasted to grow at the fastest CAGR of 9.2%, driven by stricter compliance standards and the adoption of automated, digital validation technologies
Top Companies Operating in the Bioprocess Validation Market
Tier I: Market Leaders
These companies are major providers of bioprocess validation services, with wide product/service portfolios, global reach, and strong R&D / regulatory alignment.
| Company | Key Offerings |
| Thermo Fisher Scientific | Full spectrum validation: equipment qualification, software platforms, analytical testing, extractables & leachables, microbiology, service labs. |
| Merck KGaA | Validation solutions for GMP manufacturing, single use systems, filtration, residuals/extractables, instrumentation, global presence. |
| Sartorius AG | Upstream/downstream single use validation, AI/automation enabled platforms, filter integrity, media, process qualification & continued process verification. |
Tier II: Established & Strong Regional/Niche Players
These firms are strong contributors, often with specialization or strong regional presence, especially in certain test types or geographies.
| Company | Key Offerings |
| Eurofins Scientific | Sterility & microbial detection, contract validation services, proficiency testing, regulatory support especially in the EU. |
| Pall Corporation (Danaher) | Process component validation including filters, single use & reusable systems; integrity testing and services. |
| SGS S.A. | Audit, certification, testing labs, regulatory compliance validation services across multiple regions. |
Tier III: Emerging, Niche or Smaller Players
These are companies with smaller or more specialized segments, regional players, or emerging service providers.
| Company | Key Offerings |
| Lonza Group | Validation for cell & gene therapy, viral safety, continuous bioprocess systems. |
| WuXi AppTec / WuXi Biologics | Outsourced validation, contract development & testing for biosimilars / vaccines. |
| Charles River Laboratories | Analytical method validation, extractables/leachables, toxicology / viral clearance support. |
| Others (Cobetter Filtration, DOC S.r.l., Meissner Filtration Products, Toxikon, etc.) | Specialized filtration, integrity / extractables test kits, regional labs. |
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/7030
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344
- Battery Swapping Market Size to Touch USD 24.74 Billion by 2035 - March 5, 2026
- Dental Air Polishing System Market Size to Reach USD 1603.54 Million by 2035 - March 4, 2026
- Cryoballoon Ablation System Market Size to Surpass USD 2.49 Billion by 2035, Propelled by 5.70% Growth in Minimally Invasive Cardiac Care - March 2, 2026
