CAR T-Cell Therapy Market to Hit $128.55 Billion by 2034
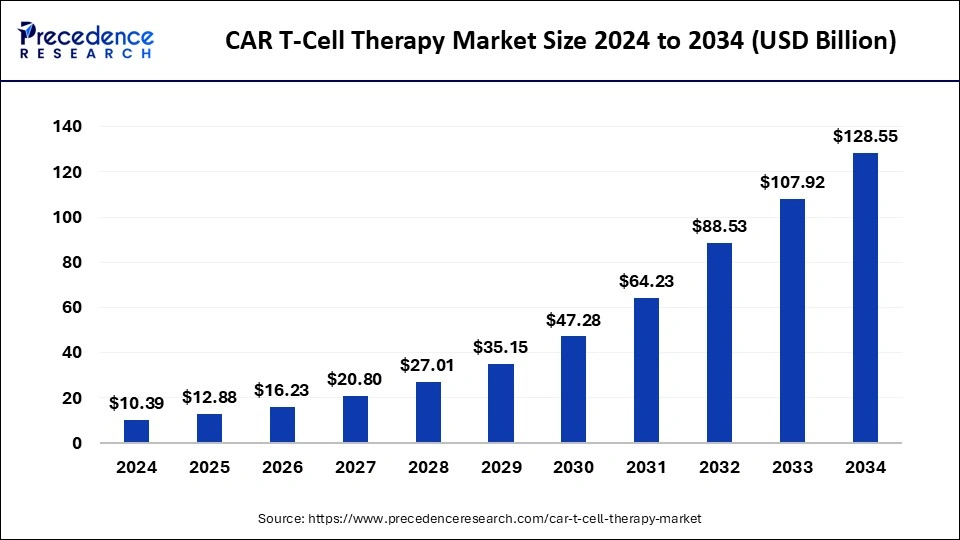
The global CAR T-cell therapy market was valued at USD 10.39 billion in 2024 and is projected to rise from USD 12.88 billion in 2025 to USD 128.55 billion by 2034, registering a robust CAGR of 29.10% (2025–2034). Growth is propelled by rising hematologic cancer incidence, rapid regulatory momentum, expanding indications (including early progress in solid tumors), and scale-up of enabling technologies—from viral vectors and dual-antigen constructs to AI-enabled R&D and manufacturing.
CAR T-Cell Therapy Market Key Highlights
- The global CAR T-cell therapy market was valued at USD 10.39 billion in 2024.
- It is projected to reach USD 128.55 billion by 2034.
- The market is expected to grow at a CAGR of 29.10% from 2025 to 2034.
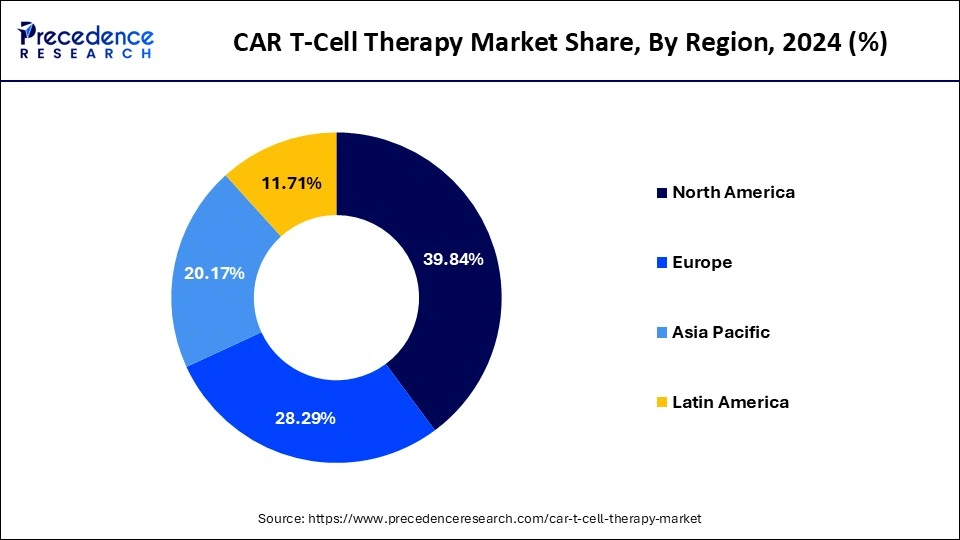
- North America dominates the market with revenue share of 39.84% in 2024.
- Asia Pacific is expected to grow at the fastest CAGR of 29.8% during the period.
- By target antigen, the CD19 segment led the market share of 45.30% in 2024.
- By target antigen, the BCMA (B-cell maturation antigen) segment is expected to grow at a significant CAGR of 27.60% over the projected period.
- By indication/disease, the hematologic malignancies segment captured the largest market share of 81.10% in 2024.
- By indication/disease, the solid tumors segment is anticipated to grow at a significant CAGR of 30.20% from 2025 to 2034.
- By type of therapy, the autologous CAR-T cell therapy segment held the biggest market share of 88.90% in 2024.
- By type of therapy, the allogeneic CAR-T cell therapy segment is expanding at a significant CAGR of 35.40% from 2025 to 2034.
- By technology/vector, the viral vectors segment dominated the market with the largest share of 76.50% in 2024.
- By technology/vector, the dual and multiple antigen targeting CAR T-cell segment is expected to grow at a significant CAGR of 33.70% from 2025 to 2034.
- By manufacturing method, the centralized manufacturing segment led the market with a major market share of 67.80% in 2024.
- By manufacturing method, the in vivo car t therapy segment is expected to grow at a significant CAGR of 36.80% over the projected period.
- By end user, the hospitals segment held a significant market share of 49.20% in 2024.
- By end user, the contract development and manufacturing organization (CDMO) segment is expected to grow at a significant CAGR of 29.50% from 2025 to 2034.
CAR T-Cell Therapy Market Size Analysis (USD Million) 2022 to 2024
CAR T-Cell Therapy Market Revenue, By Drug Type, 2022 to 2024 (USD Million)
| Drug Type | 2022 | 2023 | 2024 |
| Axicabtagene Ciloleucel | 1,118.3 | 2,472.3 | 3,046.7 |
| Tisagenlecleucel | 971.1 | 2,143.5 | 2,637.3 |
| Brexucabtagene Autoleucel | 814.8 | 1,808.5 | 2,237.5 |
| Others | 922.8 | 2,020.3 | 2,465.2 |
CAR T-Cell Therapy Market Revenue, By Indication, 2022 to 2024 (USD Million)
| Indication | 2022 | 2023 | 2024 |
| Lymphoma | 1,881.0 | 4,137.9 | 5,073.9 |
| Acute Lymphocytic Leukemia | 1,454.4 | 3,223.1 | 3,981.5 |
| Others | 491.6 | 1,083.6 | 1,331.4 |
CAR T-Cell Therapy Market Revenue, By End User, 2022 to 2024 (USD Million)
| End User | 2022 | 2023 | 2024 |
| Hospitals | 2,076.4 | 4,608.8 | 5,702.2 |
| Cancer Treatment Centers | 1,750.6 | 3,835.8 | 4,684.6 |
Why is this market important?
CAR T-cell therapy has transformed outcomes in relapsed/refractory hematologic cancers where conventional options often fail. By engineering a patient’s (autologous) or donor’s (allogeneic) T-cells to recognize tumor antigens (e.g., CD19, BCMA), CAR T can deliver deep, durable responses—including complete remissions—in high-risk populations. As platforms evolve (dual-targeting, armored CARs, safety switches), the field is widening to earlier lines of therapy and exploratory solid tumor indications. Economically, the market’s scale-up catalyzes a full-stack ecosystem—vectors, cell processing, analytics, logistics, and post-acute care—unlocking new value pools for biopharma, CDMOs, and health systems.
Key Growth Drivers
One of the most significant growth drivers is the rising global incidence of cancer, particularly blood cancers such as lymphoma, leukemia, and multiple myeloma. Conventional therapies often fail in advanced stages, leaving CAR T-cell therapy as a viable solution with higher response rates.
In addition, regulatory bodies worldwide are accelerating approvals and label expansions, improving accessibility and boosting physician confidence in these therapies. Technological innovation is another critical driver. Advancements in dual-antigen targeting, armored CAR T-cells, and in vivo delivery methods are extending applications to solid tumors, which remain an untapped growth frontier.
Finally, the integration of AI and automation into R&D and manufacturing has begun to streamline production processes, reduce human error, and enhance patient monitoring. AI-powered platforms enable real-time detection of treatment complications, optimize manufacturing consistency, and help design more effective CAR constructs.
Benefits of CAR T-Cell Therapy
CAR T-cell therapy offers several key benefits that distinguish it from traditional treatments. It provides high response and remission rates, particularly in hard-to-treat hematologic malignancies. Autologous therapies, which are tailored to individual patients, reduce the risk of immune rejection and demonstrate strong efficacy in relapsed or refractory cases. Meanwhile, allogeneic therapies present opportunities for off-the-shelf solutions, potentially lowering costs and speeding up access for critical patients.
Another important benefit is its precision targeting mechanism. CAR T-cells bind to specific antigens such as CD19 or BCMA, minimizing systemic toxicity compared to conventional chemotherapy. This targeted approach not only improves survival outcomes but also reduces long-term side effects, which is increasingly valued in personalized medicine.
What does the future hold?
Expect rapid indication expansion within hematologic malignancies, exploratory combination regimens (e.g., with checkpoint inhibitors, oncolytic viruses), and stepwise progress in solid tumors via dual/multi-antigen and microenvironment-shaping approaches. Allogeneic “off-the-shelf” and in vivo CAR T strategies will compress cost and turnaround times, while digital twins, AI-driven CMC, and real-world evidence will sharpen patient selection and lifecycle management. By the late 2020s, payers are likely to adopt outcomes-based contracts more widely, aligning reimbursement with real-world durability.
Regional Outlook
Why Did North America Dominate the CAR T-Cell Therapy Market in 2024?
North America continues to dominate the global CAR T-cell therapy market, holding 39.84% of total revenue in 2024. This leadership stems from well-established healthcare systems, a strong base of oncology specialists, and significant investment in cell and gene therapy research. The region also benefits from dense clinical trial networks and early adoption of FDA-approved CAR T products, which have been integrated into treatment protocols across top cancer centers. Academic–industry collaborations and government support for cutting-edge biotechnology further strengthen the region’s position. Canada is also playing an emerging role, with increased funding for immunotherapy research and partnerships with global pharmaceutical players to expand access.
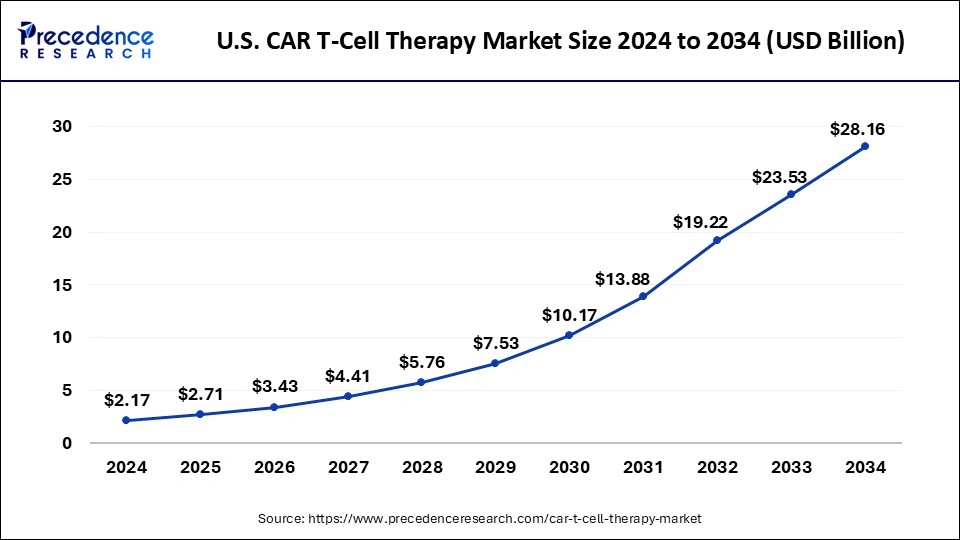
How Big Is the U.S. CAR T-Cell Therapy Market?
The United States stands as the single largest contributor within North America, showcasing the most advanced CAR T-cell ecosystem globally. The U.S. CAR T-cell therapy market was valued at USD 2.17 billion in 2024 and is projected to reach USD 28.16 billion by 2034, growing at a CAGR of 29.70%. Growth is driven by the country’s high prevalence of hematologic cancers, rising geriatric population, and rapid adoption of novel therapeutics.
Why Is Europe an Important Market for CAR T-cell therapy?
Europe holds a substantial market share, with Germany emerging as a regional leader due to its advanced healthcare facilities and proactive cancer research ecosystem. Pan-European health technology assessment (HTA) initiatives and clinical trial networks are expanding adoption, while the region’s growing elderly population is driving demand for effective cancer treatments.
Why Is Asia-Pacific the Fastest-Growing Region in the CAR T-Cell Therapy Market?
Asia Pacific is the fastest-growing market, supported by a 29.8% CAGR during 2025–2034. China leads regional growth, fueled by increasing cancer prevalence, government-backed biotech investments, and rapid clinical trial enrollment. In addition, healthcare spending is rising across the region, and local CDMOs are strengthening the manufacturing ecosystem to reduce costs and dependence on imports.
Segmentation Insights
By Target Antigen
CD19 remains the dominant target, representing 45.30% of the global market in 2024, due to its consistent expression in B-cell malignancies. BCMA, however, is the fastest-growing target, with a CAGR of 27.60%, fueled by breakthroughs in multiple myeloma treatments. Emerging targets such as CD22, HER2, and GD2 are expected to expand CAR T applications to solid tumors.
By Indication
Hematologic malignancies dominate the market with 81.10% share in 2024, owing to established clinical success in lymphomas and leukemias. However, solid tumors are expected to grow at 30.20% CAGR, as researchers focus on overcoming barriers like antigen heterogeneity and immune suppression.
By Therapy Type
Autologous therapies currently dominate with 88.90% of the market, but allogeneic therapies are set to expand rapidly due to their scalability and potential to shorten vein-to-vein times.
By Technology and Manufacturing
Viral vectors continue to lead, with a 76.50% market share in 2024. However, dual and multi-antigen CARs and in vivo delivery methods are showing the highest growth rates, reflecting innovation trends aimed at overcoming current limitations.
By End User
Hospitals are the leading end users, capturing 49.20% of the market in 2024. Their dominance is tied to robust infrastructure, specialized teams, and the ability to handle complex therapies. Meanwhile, contract development and manufacturing organizations (CDMOs) are emerging as critical players, growing at a CAGR of 29.50% as biopharma companies outsource production to manage costs and regulatory complexities.
Competitive Landscape
Market Structure: Moderately concentrated at the top with integrated leaders owning approved assets, followed by a dynamic mid-tier of clinical-stage innovators, and a long tail of platform specialists, vector suppliers, analytics firms, and service providers.

-
Leaders (Top Tier):
Novartis, Gilead Sciences (Kite), Bristol Myers Squibb, Johnson & Johnson – Global reach, approved portfolios (CD19/BCMA), active label expansions, and solid real-world datasets. -
Mid-Tier Innovators:
Allogene Therapeutics, JW Therapeutics, Curocell, Cartesian Therapeutics, Aurora Biopharma – Pushing allogeneic platforms, dual-target constructs, and new indications. -
Ecosystem & Enablers:
Lonza and other CDMOs, vector manufacturers, QC instrumentation, cryo-logistics, digital monitoring, and specialty distributors.
Ecosystem Dynamics:
-
R&D Intensity: Dual/multi-antigen and armored CARs; safety switches; next-gen vectors; in vivo delivery.
-
Access & Affordability: Outcomes-based contracts, tiered pricing, and network accreditation to spread expertise.
-
Distribution & Care Pathways: Centralized centers of excellence paired with spoke sites; tele-oncology and remote monitoring to manage toxicity and follow-up.
-
Digital Health & AI: Predictive toxicity models, automated batch release analytics, and patient-reported outcomes to refine long-term value demonstration.
AI Integration in CAR T-Cell Therapy
R&D: AI accelerates target identification, epitope mapping, and construct optimization; in silico screening narrows experimental scope.
Manufacturing: Machine vision and ML-driven controllers support closed-system automation, batch consistency, and deviation prediction—reducing cost and cycle times.
Clinical Operations: AI-enabled triage and real-time monitoring (including wearables) can flag early signs of CRS/ICANS, guiding timely interventions and potentially shortening inpatient stays.
Market Impact: Greater reliability, scalability, and cost compression—key to unlocking broader reimbursement and global access.
U.S., China, and Europe: Market Narratives
-
United States: Early adopter with leading trial density and strong reimbursement at major centers; large geriatric cohort and rising lymphoma/myeloma incidence sustain demand. Integration of AI, outcomes contracts, and decentralized pilots will shape next-stage growth through 2034.
-
China: One of the fastest-growing markets, buoyed by government support for cell and gene therapies, expanding biotech capabilities, and increased funding. Local manufacturing and regulatory agility will foster competitive pricing and regional leadership.
-
Europe (Germany Leading): High clinical sophistication and policy frameworks support uptake. Germany’s advanced centers and proactive payers create a strong foothold, with growing cross-border HTA initiatives to streamline access.
Recent Developments
-
Apr 2025 – Johnson & Johnson: EC expanded indication for Darzalex (daratumumab) SC in newly diagnosed multiple myeloma combinations, broadening treatment options for transplant-eligible and ineligible patients—supportive of the broader immuno-oncology paradigm.
-
Mar 2025 – Bristol Myers Squibb: Announced plans to acquire 2seventy bio (~USD 286 million), consolidating ownership of Abecma (BCMA CAR T for multiple myeloma) and signaling continued portfolio optimization in cell therapy.
-
Nov 2024 – FDA Approval:Obecabtagene autoleucel (Aucatzyl, Autolus) received approval for relapsed/refractory B-cell precursor ALL in adults, adding to the CD19 landscape and expanding patient options.
For inquiries regarding discounts, bulk purchases, or customization requests, please contact us at sales@precedenceresearch.com
- Battery Swapping Market Size to Touch USD 24.74 Billion by 2035 - March 5, 2026
- Dental Air Polishing System Market Size to Reach USD 1603.54 Million by 2035 - March 4, 2026
- Cryoballoon Ablation System Market Size to Surpass USD 2.49 Billion by 2035, Propelled by 5.70% Growth in Minimally Invasive Cardiac Care - March 2, 2026
