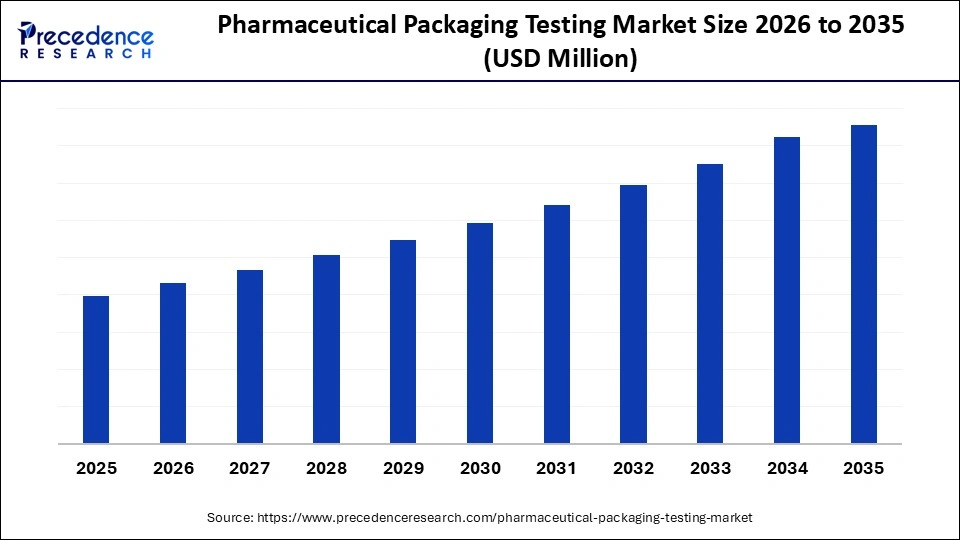
Pharmaceutical Packaging Testing Market to Reach Multimillion-Dollar Heights by 2035, Driven by Biologics Boom and AI Precision
North America Leads with Dominant Share in 2025 as Asia Pacific Charges at Fastest CAGR Through 2035 Amid Rising Global Compliance Demands
The global pharmaceutical packaging testing market surges forward, propelled by stringent regulatory mandates from FDA, EMA, and WHO ensuring drug safety across complex supply chains. With biologics, injectables, and temperature-sensitive therapies demanding flawless packaging integrity, the sector witnesses robust expansion at a projected CAGR through 2026-2035. Key drivers include escalating production of sterile injectables and the shift toward personalized medicines requiring advanced validation protocols.
Key Insights
- North America commands the largest market share in 2025, fueled by its advanced pharmaceutical ecosystem and rigorous FDA oversight.
- Asia Pacific emerges as the fastest-growing region, propelled by massive manufacturing hubs in China and India expanding biologics and vaccine output.
- Physical/mechanical testing dominates testing methods due to its critical role in verifying durability and seal integrity.
- Plastic materials lead packaging types for their versatility in bottles and blisters, while glass gains traction for sensitive injectables.
- Pharmaceutical companies hold the top end-user spot, outsourcing to contract labs for scalability. Oral solid dosage packaging captures the highest application share, with sterile injectables accelerating fastest.
Market Segmentation Breakdown
| Segment | Dominant in 2025 | Fastest Growth 2026-2035 |
|---|---|---|
| Testing Type | Physical/Mechanical | Chemical Testing |
| Packaging Material | Plastic | Glass |
| End-User | Pharma Companies | Contract Labs/Research Institutes |
| Application | Oral Drugs/Solid Dosage | Sterile Injectables/Biologics |
| Region | North America | Asia Pacific |
How Is AI Revolutionizing Pharmaceutical Packaging Testing?
Artificial intelligence transforms pharmaceutical packaging testing by deploying vision systems that detect micro-defects in seals and labels with unmatched precision, slashing human error in high-volume production. Machine learning models predict packaging failures through real-time analytics on material durability and environmental stress, enabling proactive quality control. These innovations cut validation timelines and ensure compliance for biologics demanding zero contamination.
AI-driven simulations create virtual twins of packaging under varied conditions, reducing prototype needs and accelerating market entry for injectables and vaccines. Integrated with automation, AI facilitates continuous monitoring across global supply chains, bolstering sterility assurance for sensitive therapies.
What Fuels Pharmaceutical Packaging Testing Market Growth?
Rising biologics and vaccine production mandates rigorous physical, chemical, and microbial validation to safeguard product stability during transport. Stringent global regulations amplify demand for extractables/leachables studies and container closure integrity testing (CCIT). Expansion of pharma manufacturing in emerging hubs heightens needs for compliant, scalable testing infrastructure.
What Opportunities and Trends Shape the Future?
How are biologics and cold-chain demands reshaping testing priorities?
The surge in injectables and temperature-sensitive drugs drives adoption of thermal shock and moisture barrier tests, with Asia Pacific leading via vaccine scale-ups.
What sustainability trends are emerging?
Shift to recyclable plastics and paperboard spurs environmental compatibility testing, aligning with EU circular economy goals.
How do digital tools unlock new efficiencies?
AI inspections and predictive analytics from firms like Intertek minimize recalls, while serialization combats counterfeits.
Regional Dynamics: North America Leads, Asia Pacific Surges
North America dominates in 2025, anchored by U.S. leadership in AI inspections and biologics validation amid FDA audits. Europe follows with EMA-driven sustainability focus, testing recyclability for biosimilars. Asia Pacific posts the highest CAGR, as China’s NMPA tightens GMP for exports and India’s generics boom demands CCIT scalability.
Top Companies and Breakthroughs
Leading players include SGS SA, Eurofins Scientific, Intertek Group, Nelson Labs, West Pharmaceutical Services, Gerresheimer AG, AptarGroup, Amcor plc, Berry Global, Nipro Corporation, SCHOTT AG, Catalent Inc., Thermo Fisher Scientific, Pacific BioLabs, DDL Inc., ALS Limited, Labthink Instruments, Qualitest International, PTI Packaging Technologies & Inspection, and ALMAC Group.
Recent advances: In October 2025, Intertek launched enhanced extractables/leachables services in Switzerland for Korean Pharmacopeia compliance. May 2025 saw Intertek expand its UK lab for inhaled biologics testing with advanced spectrometry. Trends highlight tamper-evident seals and smart sensors for real-time tracking.
Challenges Amid Cost Pressures
High costs of advanced testing equipment strain smaller manufacturers, especially for biologics requiring specialized sterility suites. Regulatory harmonization lags across regions, complicating global supply chains. Sustainability mandates add pressure to validate eco-materials without compromising barrier performance.
Case Study: Intertek’s Global Expansion
Intertek’s 2025 lab upgrades in Switzerland and the UK exemplify success, enabling pharma clients to navigate Korean and EU standards for injectables. This boosted their capacity for E&L and particle analysis, reducing client validation times by 30% and securing contracts with biologics leaders.
Pharmaceutical Packaging Testing Market Segments Coverged
By Testing Type/Testing Method
- Physical/Mechanical Testing
- Chemical/Chemical-compatibility/Migration/Residue/Barrier Testing
- Microbiological/Sterility/Contamination Testing
By Packaging Material Type
- Plastic (polymers) packaging
- Glass
- Paper/Paperboard/Carton/Cardboard
- Metal/Foils/Other materials
By End-User/Client/Customer
- Pharmaceutical Companies/Drug Manufacturers
- Contract Packaging/Contract Manufacturing/Contract Testing Laboratories/CROs
- Research Institutes/Testing Laboratories / QA-QC Labs
- Regulatory/Compliance/Third-party Testing Service Providers
By Application
- Sterile injectables
- Oral drug packaging
- Liquid/semi-solid therapeutics
- Biologics/specialty drugs
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East and Africa
Also Read: Pet Biotics Market Size and Growth
Get this report to explore global market size, share, CAGR, and trends, featuring detailed segmental analysis and an insightful competitive landscape overview @ https://www.precedenceresearch.com/sample/7172
You can place an order or ask any questions. Please feel free to contact us at sales@precedenceresearch.com |+1 804 441 9344
- Battery Swapping Market Size to Touch USD 24.74 Billion by 2035 - March 5, 2026
- Dental Air Polishing System Market Size to Reach USD 1603.54 Million by 2035 - March 4, 2026
- Cryoballoon Ablation System Market Size to Surpass USD 2.49 Billion by 2035, Propelled by 5.70% Growth in Minimally Invasive Cardiac Care - March 2, 2026
